You have not yet added any article to your bookmarks!

Join 10k+ people to get notified about new posts, news and tips.
Do not worry we don't spam!
Post by : Rameen Ariff
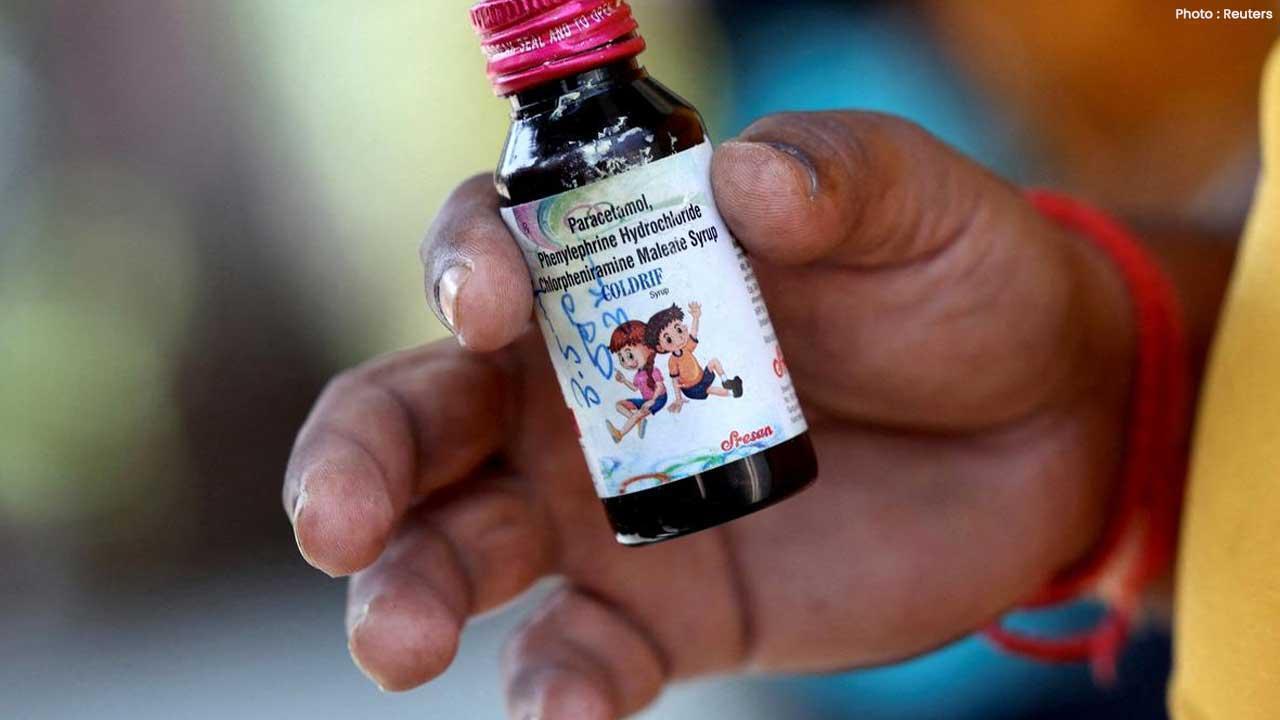
In a major breakthrough, Ranganathan Govindan, the owner of Sresan Pharmaceuticals, has been arrested in Chennai by Madhya Pradesh Police in connection with the Coldrif cough syrup tragedy that led to the deaths of several children across different Indian states. The arrest marks a significant step in uncovering one of India’s most shocking drug adulteration cases.
Authorities revealed that the toxic cough syrup, manufactured by the Tamil Nadu-based pharmaceutical company, contained diethylene glycol (DEG) — a poisonous chemical used in industrial products like glue and ink. Tests conducted earlier this month confirmed that the syrup contained DEG far beyond the safe limit, making it highly dangerous for human consumption, especially for children.
Police officials reported that the deadly syrup caused kidney failure and severe infections in many children in Madhya Pradesh and Rajasthan, with over 20 reported deaths. Following the discovery, the Tamil Nadu Drugs Control Authority suspended the company’s manufacturing license and issued a stop-production order.
After remaining on the run for several days, Govindan was tracked down and arrested during a midnight operation jointly conducted by Madhya Pradesh and Chennai police. The arrest operation began after investigators traced his vehicle movements and raided his factory in Kancheepuram, where they seized key documents and unbilled chemical containers. A transit remand has been requested to move him to Chhindwara, where most of the child deaths occurred.
According to the investigation, Sresan Pharmaceuticals was found to be illegally operating even after being removed from the Ministry of Corporate Affairs register. The company continued its production under a proprietary setup, raising serious concerns about regulatory loopholes in India’s drug manufacturing system.
Health authorities confirmed that Coldrif syrup contained 46–48% diethylene glycol, against the permitted limit of 0.1%. The toxic compound can severely damage the kidneys, liver, and nervous system, particularly in young children.
As the investigation expands, officials are now focusing on identifying all links in the supply chain, including chemical suppliers, distributors, and stockists, to determine how the tainted syrup reached pharmacies and families.
So far, at least nine states have banned Coldrif syrup, while India’s top drug regulator has admitted to lapses in manufacturing inspections, stating that many companies failed to test every batch of raw materials used in medicine production.
The Coldrif cough syrup case has once again highlighted the urgent need for strict quality control and stronger regulatory oversight in India’s pharmaceutical industry to prevent such tragedies in the future.










Australia Repatriates ISIL-Linked Families
Nineteen women and children with alleged ISIL ties returned from Syria as Australian authorities lau

Airlines Suspend Flights Amid Mideast War
Global airlines cancel and reroute flights across the Middle East as the Iran conflict disrupts avia

US-Armenia Deal Signed Before Elections
United States and Armenia signed a strategic partnership agreement as Yerevan strengthens ties with

Turkey Opposition Plans New Party Congress
CHP chairman Kemal Kilicdaroglu says party congress will be held after legal procedures are complete

Philippines Launches Drugs War Truth Panel
New independent commission will investigate alleged extrajudicial killings linked to former Presiden

Cambodia Pushes $300B Energy Plan Fast
Global fuel crisis and Strait of Hormuz tensions push Cambodia to speed up efforts to unlock dispute